|

Developers:
|

|
Ms. Virginia Corcoran
Amosland Elementary School
Ridley, PA
|
Dr. Carl Hemenway
Rohm and Haas Company
Bristol, PA
|
|
|
|
|
|
|
Grade
Levels:
|

|
3 through 6
|

|
|
|
|
|
Discipline:
|

|
General Science
|
|
|
|
|
|
Goals:
|

|
To illustrate the molecular nature of matter and to show
that chemical change involves alteration of molecules.
This activity provides a specific example of a chemical
change. It is designed to follow previous work in which the
students discuss, in general, the difference between
physical change and chemical change. This activity will work
to aid any teaching unit on chemical change, although it was
originally designed as a supplement to Windows on Science, a
videodisc-based curriculum program (Optical Data Corp.,
Warren NJ).
Vinegar reacts with baking soda to form bubbles of carbon
dioxide gas in a simple, well-known chemical reaction. The
teacher can demonstrate this reaction, and the students can
�act out� the roles the molecules play in the reaction.
Vinegar is mostly water, with a few other chemicals
dissolved in it to give it color, flavor, and acidity. The
acidity comes from a small amount of acetic acid, which has
the chemical formula CH3COOH. Most of the
CH3COOH molecules stay intact, but a few of them
separate into two parts: the hydrogen ion (H+) and the
acetate ion (CH3COO-). The acetate ion does not
participate in the reaction with baking soda, so we ignore
it in the "role playing." For this exercise, vinegar will be
a lot of water (H2O) molecules with a few
hydrogen ions wandering around among them.
Baking soda is a white powder made of a pure chemical
called sodium bicarbonate (NaHCO3). When this
powder is added to water, it separates completely. Every
molecule separates into a sodium ion (Na+) and a bicarbonate
ion (HCO3-). The sodium ion does not participate
in the reaction with vinegar, so we will ignore it in our
molecular drama. The chemical reaction is written like this:
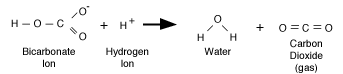
|
|
|
|
|
|
Materials:
|

|
Quart of white vinegar; small box of baking soda;500 mL
glass beaker (or sturdy canning jar); tablespoon; paper (one
sheet, 8-1/2 X 11 inches, for each student); broad marker;
tape.
|
|
|
|
|
|
Management:
|

|
This exercise consists of a brief demonstration followed
by some participatory activity. It should be complete within
a one-hour period. The participatory activity requires a
large space, so it should be performed in a gymnasium or
outdoors.
|
|
|
|
|
|
Procedure:
|

|
1. Demonstration:
The teacher should wear safety glasses while performing the
demonstration. The individual materials are nonhazardous,
and the chemical reaction is mild; but you should always
wear glasses when conducting chemical experiments.
Pour vinegar into the beaker, so that the beaker is about
half full. Add one tablespoon of baking soda to the vinegar.
Ask the students to observe what happens. Discuss how the
fizzing and bubbling are different from physical changes.
2. Molecular "Role Playing"
Move to the gym or playground. Make a "beaker" on the ground
by drawing three sides of a square, with each side about 15
feet long.
Each student will be an atom, either Hydrogen (H), Oxygen
(O), or Carbon (C). Here is a sample of how to group a class
of 25 students into molecules:
Take 3 students; use the 8-1/2 X 11-inch paper to label 2
of them as "H" and one as "O". Instruct the O student to
hold hands with the two H students, like this:
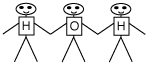
This group is a water molecule. Tell the water molecule to
keep their hands clasped, and to wander around randomly
inside the "beaker." They will continue to hold hands with
each other, but they will not clasp anybody from any other
molecules.
Make 5 more water molecules and set them all to wandering
around together in the beaker. Each molecule should walk
around aimlessly, and they should (GENTLY) bump into each
other and into the sides of the "beaker."
Pick a lone student and give a label "H." Tell that
student to go into the beaker alone and wander around. Pick
another lone H to wander around, too.
You now have "vinegar" in the beaker: mostly water, with
a few hydrogen ions around to make it an "acid."
The remaining students should be labeled with one "H,"
one "C," and three "O." They should hold hands something
like this, to form the "bicarbonate" ion:
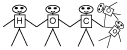
The bicarbonate group should approach the open "top" of the
beaker and start to enter the group of water molecules. As a
group, they should wander around until they get near a lone
H ion. At that time, they "react" with the H ion, like this:
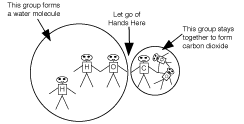
The new water molecule joins the other water molecules in
wandering around the beaker. The new carbon dioxide molecule
wanders toward the open end of the beaker and leaves the
beaker.
|
|
|
|
|
|
Closure:
|

|
In discussing the demonstration and role-playing with the
students, the teacher should be sure that the following key
points are covered:
- Matter is a collection of tiny molecules.
- Each molecule is a collection of atoms.
- In chemical changes, the molecules break apart, and
the atoms form new combinations.
- The clues that a chemical change has taken place:
color change; temperature change; phase change (new
solids, liquids, or gases). After the class discussion,
the students should retell the "story" of the chemical
reaction in their own ways. Something other than humans
should be the atoms (animals, clouds, blobs of jelly,
houses, cars, or whatever), and the story should involve
the key points of separation and regrouping. Older
students could write the story as a prose narrative;
younger ones could draw cartoons or even just "before"
and "after" pictures.
|
|
|
|
|
|
|

|
Ridley School District - Grade 3
Matter and Energy Unit - Overview
|
|
|
|
|
|
|
|
|
|
Concept 1:
|

|
Matter is anything that takes up space and has mass.
Matter can be defined by its properties.
Learning Outcome: Children will classify matter by
properties.
|
|
|
Activities:
|

|
- Sort boxes
- Language Arts tie-in (adjectives)
- Ice cube in Alcohol/Water for unseen properties
- Oobleck
|
|
|
|
|
|
Concept 2:
|

|
Matter exists in the form of solid, liquid and gas.
Learning Outcome: Children will experience matter in
its three states.
|
|
|
Activities:
|

|
- Test if air has weight using balloons
- Acrostic or collage of solid, liquid and gas
- Evaporating ice
|
|
|
|
|
|
Concept 3:
|

|
Matter is made from particles too small to be seen,
called atoms. The movement of atoms relates to the state of
matter.
Learning Outcome: Children will physically and
visually depict the atomic structure of matter in its three
states.
|
|
|
Activities:
|

|
- Use bodies to show arrangement of atoms in solids,
liquids and gases.
- Use marshmallows.
- Have SLG Treats (Ice cream sodas).
|
|
|
|
|
|
Concept 4:
|

|
Temperature is a property of matter.
Learning Outcome: Children will measure temperature
of matter.
|
|
|
Activities:
|

|
- Practice with thermometers inside and outside.
|
|
|
|
|
|
Concept 5:
|

|
Heat energy causes the movement of atoms. A conductor is
matter that allows heat to move easily, an insulator is
matter that slows the movement of heat.
Learning Outcome: Children will create effective
conductors and insulators.
|
|
|
Activities:
|

|
- Magic trick using pennies
- Testing materials in heat
- Great ice cube contest (making insulators)
- Is your hat a good insulator
|
|
|
|
|
|
Concept 6:
|

|
Changes in matter when heated or cooled.
Learning Outcome: Children will demonstrate the
following changes in matter: condensation, evaporation,
melting, freezing, contraction and expansion.
|
|
|
Activities:
|

|
- Task cards for cooperative groups to work at
stations.
- The great meltdown
- Paint with melted crayons.
|
|
|
|
|
|
Concept 7:
|

|
Physical change is a change in matter without changing
the atoms. Chemical change is a change in matter in which a
completely new type of matter is formed.
Learning Outcome: Children will create chemical
change and will be able to differentiate chemical change
from physical change.
|
|
|
Activities:
|

|
- Stations set up for cooperative groups.
- Vinegar and baking soda reaction using bodies as
atomic model.
|
|
|
|
|
|
Concept 8:
|

|
People physically and chemically change matter to make it
more useful.
Learning Outcome: Children will be able to change
matter physically and chemically to make a new type of
matter.
|
|
|
Activities:
|

|
- Lemon fizz
- Experimenting with the amount of baking powder in
pancakes and cupcakes.
- Invent-a-new-use. Recycling or physically changing
used matter to make something new.
|
|
|
|
|
|
|

|
Chemistry in Cooking
Some chemical changes form gases as a product, as in the
case of baking soda and vinegar. When the reaction takes
place in a liquid, the bubbles rise to the surface and can
be easily observed. When the reaction takes place in a
batter that is being warmed, the bubbles get trapped and are
permanently set. This reaction makes baked goods airy and
soft instead of hard and flat.
Try these two recipes with your class to experiment with
the effects of baking powder in foods.
Divide the class up into three groups with each group
adding a different amount of baking powder to the recipe.
Basic Cupcake Recipe
This recipe makes 4 cupcakes. Group 1 will add 1/2 tsp of
baking powder to the dry ingredients, Group 2 will add 1
tsp., and Group 3 will add 1-1/2 tsp. of baking powder.
1 cup flour
1/2 cup sugar
1 egg
1/4 cup butter (room temp)
1/4 cup milk
1 tsp vanilla
? baking powder
Mix dry ingredients. Each group adds the required amount
of baking powder. In another bowl mix butter, eggs, milk and
vanilla. Add wet ingredients to dry and mix until flour is
moist. Beat till smooth.
Bake in a 350 degree oven for 15 minutes. Compare and
enjoy. You will notice differences among the cupcakes in
crumb size, height, and taste.
Pancakes
For a breakfast treat for your class, make pancakes. This
recipe yields 12 pancakes. Vary the amount of baking powder
with each batch.
1-1/2 cups flour
1 tsp salt
3 tbsp sugar
1 lightly beaten egg
3 tbsp melted butter
1-1/4 cup milk
1 tsp baking powder (Batch 1)
1-3/4 tsp. baking powder (Batch 2)
2-1/4 tsp. baking powder (Batch 3)
Use a few quick strokes to moisten dry ingredients. Don't
overbeat.
|



